Here’s what’s in store for you in today’s issue:
🍄 The cure for the worst pain known to humans
🍄 Combining antidepressants and psilocybin
🍄 More positive findings from biggest psilocybin trial
🍄 Psychedelic cream?
🍄 And more.
Be sure to check out this week’s Daily Mushroom podcast:
Why a psychedelic retreat might be the best vacation you’ve ever been on
In this episode of the Daily Mushroom Podcast, we have the CEO of Silo Wellness, Douglas Gordon. Silo Wellness offers legal, psychedelic retreats in Jamaica and Oregon. Their mission is heavily rooted in the education around the physical and mental benefits of mushrooms. We hope you enjoy this one!

Antidepressants + psilocybin
Do patients need to come off of antidepressants before trying psilocybin therapy?
New research from MindMed (MNMD) and Liechti Labs suggests that it’s not necessary – and there are actually benefits of combining the two.
Escitalopram (aka Lexapro) did not affect the way psilocybin was processed and helped reduce negative side effects like anxiety and high blood pressure.
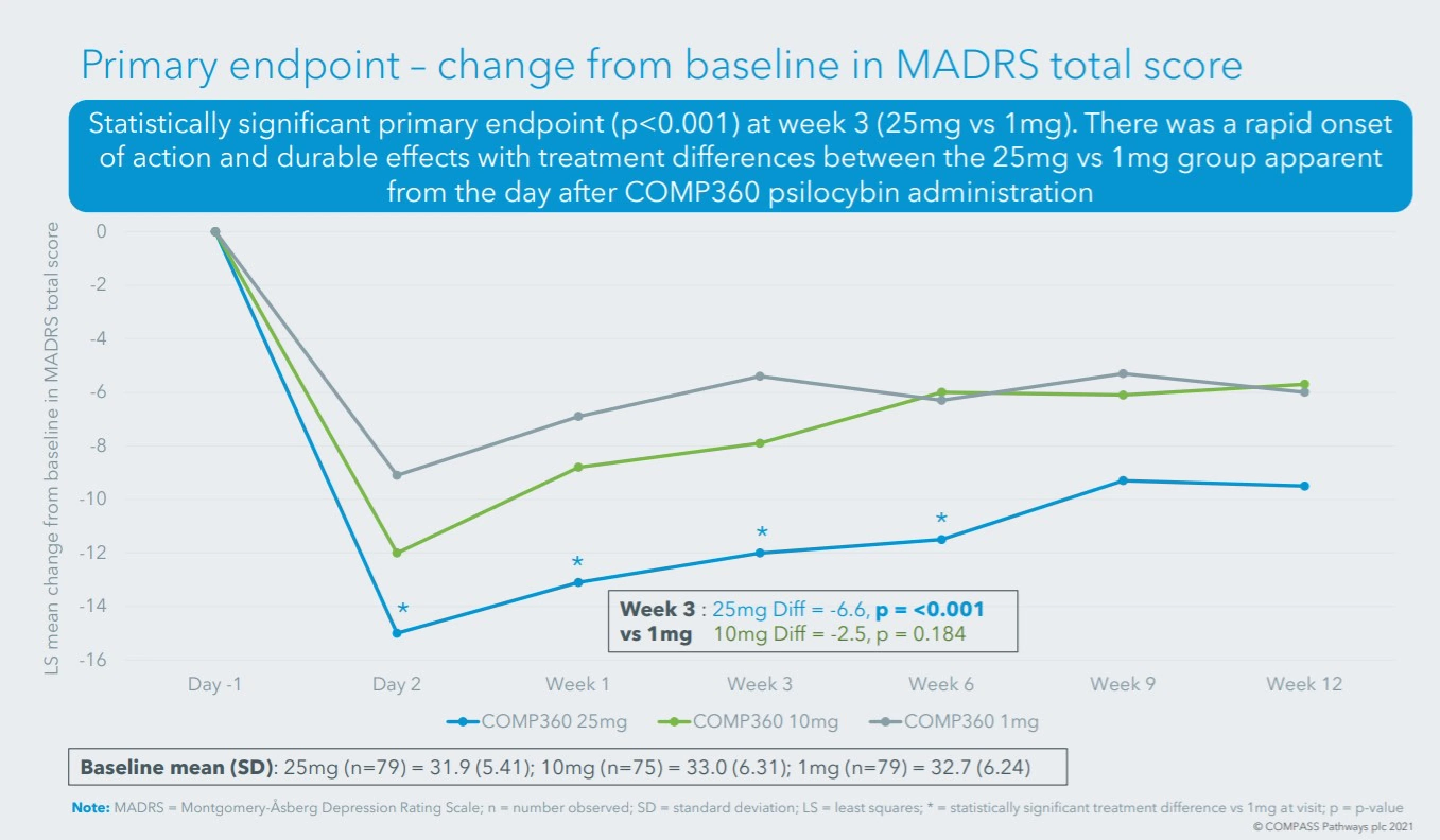
More positive findings from biggest psilocybin trial
COMPASS Pathways (CMPS) announced additional findings from the world’s largest psilocybin trial for depression.
In addition to reduced depression scores, patients showed improvements in anxiety, daily functioning, cognition, and quality of life. Just one day after the treatment, patients had increased positive feelings such as “interested, excited, strong” and decreased negative feelings like “distressed, upset, afraid.”
Psychedelic cream
Psycheceutical Inc., a private Florida company, is developing a unique way to deliver psychedelics – through a cream or patch applied to the back of the neck. This method allows psychedelics to enter the brain in just 5-10 minutes without entering the bloodstream, which reduces negative side effects.
Participate in psychedelic research from your phone!
Quantified Citizen is a platform made in collaboration with Paul Stamets to accelerate health research. It allows anyone to participate in studies anonymously from their phone.
The platform was used to conduct the world’s largest mobile microdosing study, which revealed that microdosers exhibit lower levels of depression, anxiety, and stress.
It’s not too late to participate – download the app here to join the study!

Curing the worst pain known to humans
For years, Brad Badelt suffered from debilitating cluster headaches (which are often called “suicide headaches” and described as the worst pain known to humans.
After seeing countless neurologists, chiropractors, acupuncturists, and naturopaths, he would still experience the horrible headaches several times a day for weeks on end.
After taking one dose of shrooms, things finally changed.
“When I woke the next morning, I felt different: the mental fog brought on by my cluster headaches was gone. For the next twelve months, I remained headache free. Every year since, I’ve downed a dose of shrooms, and every year my cluster headaches have remained in remission.”
Want a life-changing psychedelic trip but don’t know where to start?
The Wakeful Integration Journal is your guide to transformation.
It’s the ultimate tool to help you prepare for, navigate, and deeply integrate your psychedelic experiences. The journal features:
- Prompts to discover your intentions and reflect on your trip
- Dose tracking
- Tips to support you along the journey
- Colouring pages to unlock your creativity
If you want to start with microdosing rather than a full-on trip, the Wakeful Intention Journal is for you! It guides you through a 6-week microdosing protocol and helps you set intentions, notice patterns, and integrate your insights.
Support the Kickstarter here to receive a journal and begin your transformation journey!
The Canadian government is funding psychedelic research!
MYND Life Sciences (MYND) received $45,600 in government funding to support a psilocybin research project on treatment resistant depression and neuroinflammation. The project will run now until March 2022.

Novamind (NM) partnered with Bienstar Wellness to develop the first network of integrative mental health clinics in Latin America. The companies plan to bring psychedelic medicine to Uruguay, Brazil, Mexico, Peru, and Panama.
PharmaTher (PHRM) was granted a Japanese patent for KETBET, a combination of ketamine and betaine anhydrous that can be used for various mental health and pain disorders. Betaine anhydrous enhances the antidepressant effect of ketamine while reducing the side effects.
Telescope Innovations (TELI) acquired the rights to the University of British Columbia’s research on synthetic psilocybin in exchange for 1M common shares at $0.99 each.
Bright Minds Biosciences (DRUG) announced that its drug discovery program has synthesized hundreds of patentable psychedelic compounds. Researchers are optimizing the most encouraging molecules and aim to announce the lead molecule early next year.
Shanti Therapeutics, a subsidiary of Global Wellness (GWS), is awaiting ethics committee approval to conduct one of the first studies on MDMA for pain management.
These companies announced their financial highlights:

Here’s what you missed in the legal world:
- New Zealand is legalizing drug testing so users can check the purity of illegal substances. The services will be available at events like concerts and festivals. Read more…
- The DEA is once again increasing the production quotas of these psychedelic drugs for 2022:
- Psilocybin increased from 3,000 to 8,000 grams
- Psilocin increased from 2,000 to 3,000 grams
- MDMA increased from 3,200 to 8,200 grams
- DMT increased from just 250 to 3,000 grams
The new limits will allow more researchers to study the drugs! Read more…
A Florida Rep. filed a bill to decriminalize all illegal substances and implement rehabilitative health intervention or civil fines instead. Read more…

Eco-friendly funerals
A startup in the Netherlands is making biodegradable “living coffins” out of mushroom mycelium!
Not only do the coffins enrich the soil, but they allow “people to become one with nature again.”
After months of testing, the first funeral using a mushroom coffin was a success.

Bob Marley mushrooms 🇯🇲
Silo Wellness (SILO) launched a line of functional mushroom products in collaboration with Bob Marley’s family!
The Marley One line features:
- One Mind: A coffee-flavored blend of lion’s mane and gingko biloba designed to improve focus and cognitive function.
- One Flow: A peppermint-flavored blend of cordyceps and ginseng designed to enhance physical endurance and mental function.
- One Harmony: A mango-flavored blend of chaga and ginger designed to stimulate gut health and improve digestion.
- One Body: A berry-flavored blend of turkey tail and astragalus designed to support immune health.
- One Rest: A vanilla-flavored blend of reishi and GABA designed to help reduce tension and stress and improve quality of sleep.
Use promo code DAILYMUSHROOM20 to get 20% off!

Industry Quick Hits
Nov 29 – Numinus (NUMI) Develops Proprietary Psychedelic Tests with New Toxicity and Potency Scans Read more…
Nov 29 – atai Life Sciences (ATAI) Increases its Ownership Position in COMPASS Pathways (CMPS) Read more…
Nov 30 – Cybin (CYBN) Announces FDA IND and IRB Approvals for Clinical Trial to Treat Frontline Clinicians with COVID-Related Distress Read more…
Dec 2 – Ketamine One (MEDI) Partners with Cognetivity Neurosciences in Collaborative Clinical Study on Depression and PTSD Read more…
Dec 2 – Tryp Therapeutics (TRYP) Receives Confirmation from FDA to Proceed with Phase 2a Study in Fibromyalgia Read more…
Nov 26 – Delic Corp’s (DELC) Ketamine Wellness Centers Opening Salt Lake City & Reno Locations Read more…
Video of the Week
Playlist of the Week