After his brother overdosed in 2019, Timothy Ko founded Entheon Biomedical (ENBI) on his quest to find a better treatment for addiction through psychedelic therapy.
The company is focused on advancing the DMT molecule to treat substance use disorders. It just submitted a regulatory package to an ethics committee for a human clinical trial on healthy smokers, which will be one of the most comprehensive studies of DMT ever.
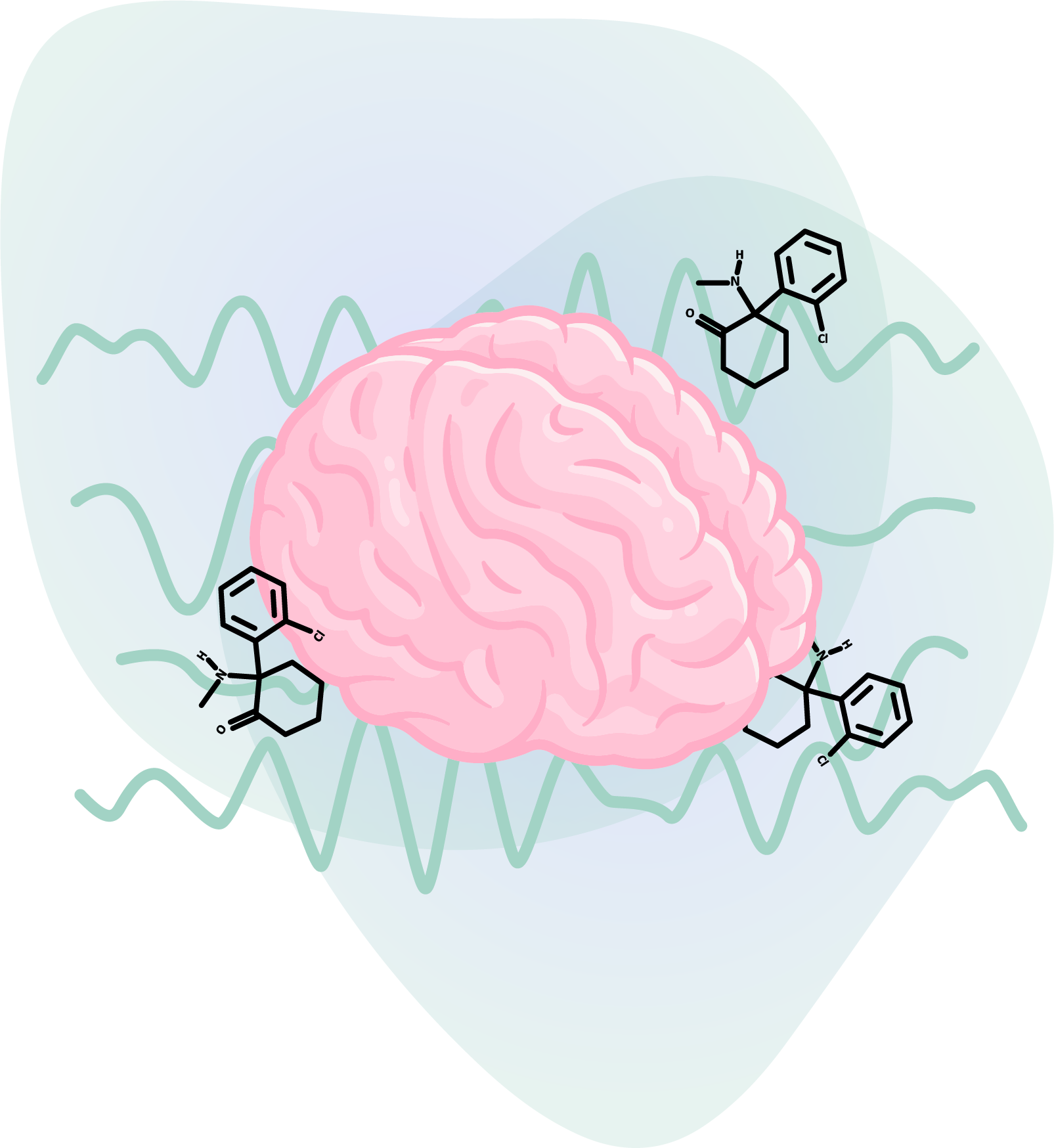
Entheon has also begun recruiting patients with treatment-resistant depression for a ketamine therapy study, which will be conducted with Heading Health LLC. The study will use an electroencephalography (EEG) headset and genetic screening to:
- Understand how ketamine influences brainwave patterns
- Investigate how genetic markers influence ketamine response
- Explore the effects of music on therapeutic outcomes
- Develop a framework for characterizing the psychedelic drug state